
The Urea Breath Test (UBT) for Helicobacter pylori (H. pylori) is widely used in clinical and health management due to its non-invasive nature, high accuracy, and convenience.
Introduction to the Helicobacter pylori Urea Breath Test
Helicobacter pylori (H. pylori) is a common pathogenic bacterium in the stomach, closely associated with diseases such as gastritis, gastric ulcers, duodenal ulcers, and gastric cancer. The Urea Breath Test (UBT) is a non-invasive, rapid, and accurate diagnostic method used to detect H. pylori infection.
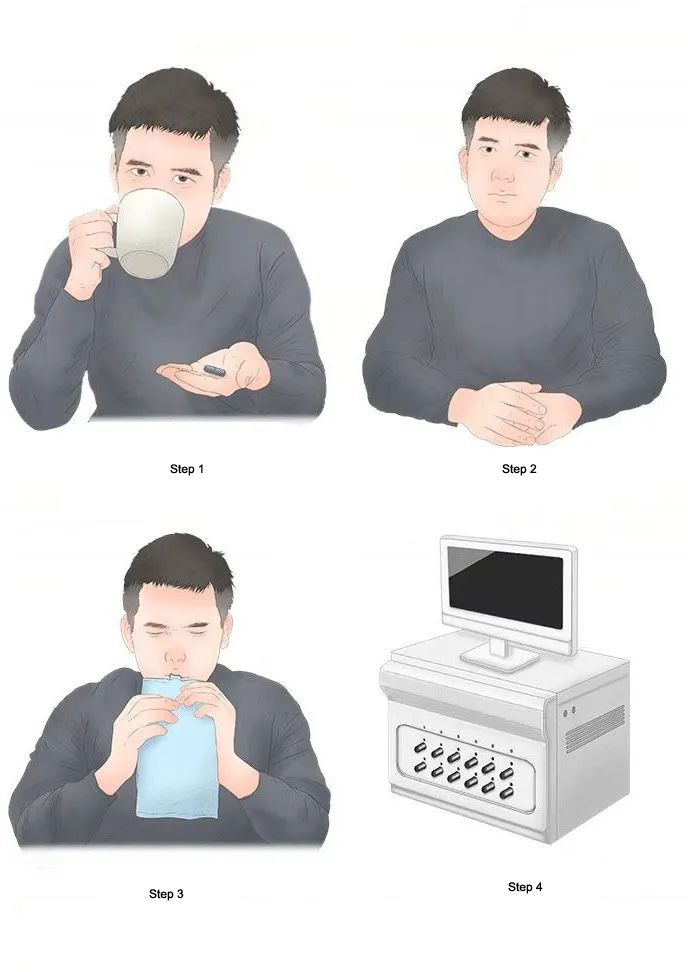
Detection Principle
Characteristics of Helicobacter pylori:
H. pylori could produce urease, which breaks down urea into ammonia and carbon dioxide (CO₂).
Detection Process:
1.The patient orally ingests a urea reagent containing labeled carbon (¹³C or ¹⁴C).
2.If H. pylori is present in the stomach, the urease will decompose the labeled urea, producing labeled CO₂.
3.The labeled CO₂ is absorbed into the bloodstream and subsequently exhaled through the lungs.
4.By collecting the patient's breath sample and analyzing the labeled CO₂ content, the presence of H. pylori infection can be determined.
Advantages of the Test
1.Non-invasive: No endoscopy or biopsy is required, ensuring a more comfortable patient experience.
2.High Accuracy: Sensitivity (>95%) and specificity (>95%) are both high, providing reliable results.
3.Rapid and Convenient: The test is quick, typically taking only about 30 minutes.
4.High Safety: The use of non-radioactive labeled carbon (¹³C) makes it suitable for special populations such as children and pregnant women.
5.Wide Applicability: Can be used for initial diagnosis, treatment efficacy evaluation, and recurrence monitoring.
Target Population
1.Patients with symptoms such as indigestion, stomach pain, or acid reflux.
2.Patients suspected of having gastritis, gastric ulcers, or duodenal ulcers.
3.Individuals with a family history of gastric cancer or long-term H. pylori infection at high risk.
4.Patients requiring evaluation of H. pylori eradication treatment efficacy.
5.Patients unsuitable for or unwilling to undergo endoscopy.
Procedure
Pre-test Preparation:
1.The patient must fast (typically for 4-6 hours).
2.Discontinue antibiotics, proton pump inhibitors (PPIs), and other medications for at least 2 weeks to avoid false-negative results.
Test Steps:
1.The patient orally ingests the urea reagent containing labeled carbon.
2.Wait for 15-30 minutes to allow the urea to fully react with H. pylori in the stomach.
3.Collect the patient's breath sample (usually using a breath bag or specialized device).
4.Analyze the labeled CO₂ content in the breath sample using a mass spectrometer or infrared spectrometer.
Result Interpretation:
If the labeled CO₂ content exceeds the threshold, the result is positive for H. pylori; otherwise, it is negative.
Clinical Application Scenarios
1.Initial Diagnosis and Screening:
Used for the preliminary diagnosis of patients with gastrointestinal symptoms to quickly determine the presence of H. pylori infection.
2.Treatment Efficacy Evaluation:
Used to assess the effectiveness of H. pylori eradication treatment (typically performed 4 weeks after treatment completion).
3.Recurrence Monitoring:
Regularly monitor patients with a history of H. pylori infection for recurrence.
4.Epidemiological Surveys:
Used for large-scale population surveys to determine H. pylori infection rates.
5.Special Population Testing:
Suitable for children, pregnant women, the elderly, and others unsuitable for endoscopy.
Precautions
1.Medication Interference: Antibiotics, bismuth agents, and PPIs may affect test results and should be discontinued in advance.
2.Dietary Interference: Avoid high-protein foods or carbonated beverages before the test.
3.Equipment Requirements: Professional analytical instruments are required to ensure accurate results.
The Urea Breath Test (UBT) for Helicobacter pylori (H. pylori) is widely used in clinical and health management due to its non-invasive nature, high accuracy, and convenience. Below are its main application scenarios and specific cases:
I. Application Scenarios
1.Initial Diagnosis and Screening
Scenario Description: Patients presenting with symptoms such as indigestion, stomach pain, acid reflux, and nausea, where H. pylori infection is suspected, require initial diagnostic testing.
Case Example: A 35-year-old male patient with long-term stomach discomfort underwent a UBT, which returned a positive result, confirming H. pylori infection. He was subsequently treated with antibiotics.
2.Treatment Efficacy Evaluation
Scenario Description: After completing H. pylori eradication therapy, patients need to be assessed to confirm successful bacterial clearance.
Case Example: A 50-year-old female patient underwent a UBT four weeks after completing triple therapy (antibiotics + proton pump inhibitor). The negative result indicated successful treatment.
3.Recurrence Monitoring
Scenario Description: Patients with a history of H. pylori infection require regular monitoring for recurrence, especially if symptoms reappear.
Case Example: A 60-year-old male patient, previously treated for H. pylori-induced gastric ulcer, experienced stomach pain again one year later. A UBT confirmed recurrence with a positive result.
4.Screening in High-Risk Groups for Gastric Cancer
Scenario Description: Individuals with a family history of gastric cancer or long-term H. pylori infection are screened regularly to prevent gastric cancer.
Case Example: A 45-year-old female with a family history of gastric cancer was advised to undergo annual UBTs. Early detection and treatment of H. pylori infection helped reduce her gastric cancer risk.
5.Testing in Children and Pregnant Women
Scenario Description: UBT is a safe and preferred method for special populations, such as children and pregnant women, who are unsuitable for endoscopy.
Case Example: An 8-year-old child with recurrent abdominal pain underwent a UBT, which returned a positive result. The child was diagnosed with H. pylori infection and treated with a pediatric-specific regimen.
6.Epidemiological Surveys
Scenario Description: UBT is used for large-scale population surveys to assess H. pylori infection rates in specific regions or groups.
Case Example: A regional H. pylori screening program tested 1,000 residents using UBT, revealing a 40% infection rate. This data supported the formulation of public health policies.
7.Preoperative Screening
Scenario Description: Patients scheduled for gastric surgery are screened for H. pylori infection to reduce postoperative complications.
Case Example: A patient requiring endoscopic surgery for gastric polyps tested positive for H. pylori during preoperative UBT. Eradication therapy was administered before surgery to minimize infection risks.
II. Case Studies
Case 1: Diagnosis and Treatment of Dyspepsia
Patient Background: A 28-year-old female with long-term post-meal bloating, acid reflux, and occasional stomach pain.
Testing Process: UBT returned a positive result, confirming H. pylori infection.
Treatment Plan: A 14-day triple therapy (antibiotics + proton pump inhibitor) was administered.
Efficacy Evaluation: A follow-up UBT four weeks post-treatment showed a negative result, and symptoms completely resolved.
Case 2: Early Intervention in High-Risk Gastric Cancer Patient
Patient Background: A 52-year-old male with a family history of gastric cancer, asymptomatic, advised to undergo H. pylori screening during a routine check-up.
Testing Process: UBT returned a positive result.
Treatment Plan: Eradication therapy was initiated, with annual follow-up UBTs recommended.
Long-Term Management: Early intervention reduced the risk of gastric cancer development.
Case 3: Diagnosis of Abdominal Pain in a Child
Patient Background: A 10-year-old boy with recurrent abdominal pain, raising parental concerns about gastric issues.
Testing Process: UBT returned a positive result.
Treatment Plan: A pediatric-specific antibiotic regimen was administered.
Efficacy Evaluation: Symptoms resolved post-treatment, and a follow-up UBT showed a negative result.
Case 4: Prevention of Postoperative Infection
Patient Background: A 45-year-old female requiring gastric surgery for a gastric ulcer.
Testing Process: Preoperative UBT returned a positive result.
Treatment Plan: Eradication therapy was administered before surgery.
Outcome: Postoperative UBT showed a negative result, and no infection-related complications occurred.
III. Summary
The Urea Breath Test for H. pylori is invaluable in the following scenarios:
Initial Diagnosis and Screening: Rapid detection of H. pylori infection.
Treatment Efficacy Evaluation: Confirmation of successful eradication.
Recurrence Monitoring: Regular monitoring for infection recurrence.
High-Risk Group Screening: Prevention of severe conditions like gastric cancer.
Special Population Testing: Safe for children, pregnant women, and others unsuitable for endoscopy.
Epidemiological Surveys: Provides data for public health policy formulation.
Through real-world cases, it is evident that UBT has extensive clinical applications, enabling rapid and accurate diagnosis and management of H. pylori infection, ultimately improving patient outcomes.